Chemistry: Solutions (Ionic and Molecular) 2nd, 1983 (silent)
Chemistry: Solutions (Ionic and Molecular) 2nd, 1983 (silent)
Overview
This educational segment explores the scientific study of ionic and molecular solutions, utilizing visual aids to effectively illustrate fundamental concepts. It begins by introducing the basic principles of chemical solutions, showcasing how different elements interact and combine to form solutions, as well as detailing the molecular structure of water. The differences between ionic and molecular structures are clearly highlighted, emphasizing their representation and concentration within solutions.
Additionally, a laboratory experiment is featured, demonstrating the principles of solubility through the interaction of carbon tetrachloride (CCl₄) and oil. This experiment is presented with clear labeling to enhance understanding, providing viewers with a practical illustration of the behavior of solutions and the factors influencing solubility. Overall, the segment serves as a comprehensive exploration of the characteristics and behaviors of ionic and molecular solutions.
Email us at footage@avgeeks.com if you have questions about the footage and are interested in using it in your project.
#Ionic Solutions #Molecular Solutions #Chemical Reactions #Solubility Experiment #Visual Learning
Timeline
00:00:00 – 00:04:00 Introduction to Ionic and Molecular Solutions
00:01:20
The video begins with a scientific exploration of solutions, focusing on ionic and molecular types, presented with visual aids but no audio.
00:04:00 – 00:08:00 Introduction to Chemical Solutions
00:07:06
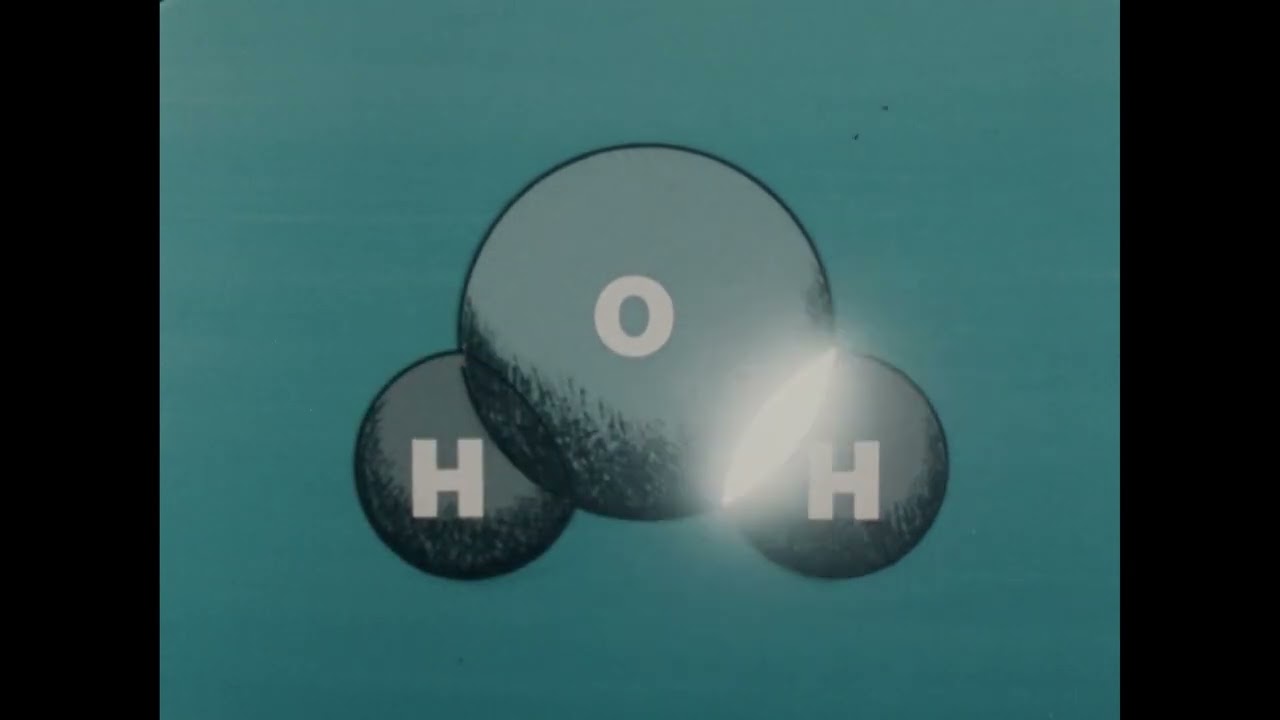
The segment begins with a visual representation of chemical solutions, focusing on how hydrogen atoms and other elements combine to form various solutions.
00:07:15
An animation further illustrates the molecular structure of water, showing one oxygen atom bonded to two hydrogen atoms, emphasizing its role as a solvent in chemical processes.
00:08:00 – 00:12:00 Ionic and Molecular Structures
00:08:51
The scene visually represents ionic and molecular structures, demonstrating interactions between different sized particles in a solution.
00:10:40
The movement of tree branches and leaves against the sky could metaphorically represent the dissolution process or the dispersal of molecules in a solution.
00:11:34
The segment displays bottles of sulfuric acid and hydrochloric acid, comparing their dilute and concentrated forms to highlight differences in their ionic and molecular structures.
00:12:00 – 00:22:41 Carbon Tetrachloride Experiment
00:18:57
A beaker of clear carbon tetrachloride (CCl
₄) is displayed in a laboratory setting, likely for a chemistry demonstration.
00:20:50
An experiment shows oil interacting with carbon tetrachloride (CCl
₄) in a beaker, illustrating solubility and solution behavior with labeled cards for clarity.










